 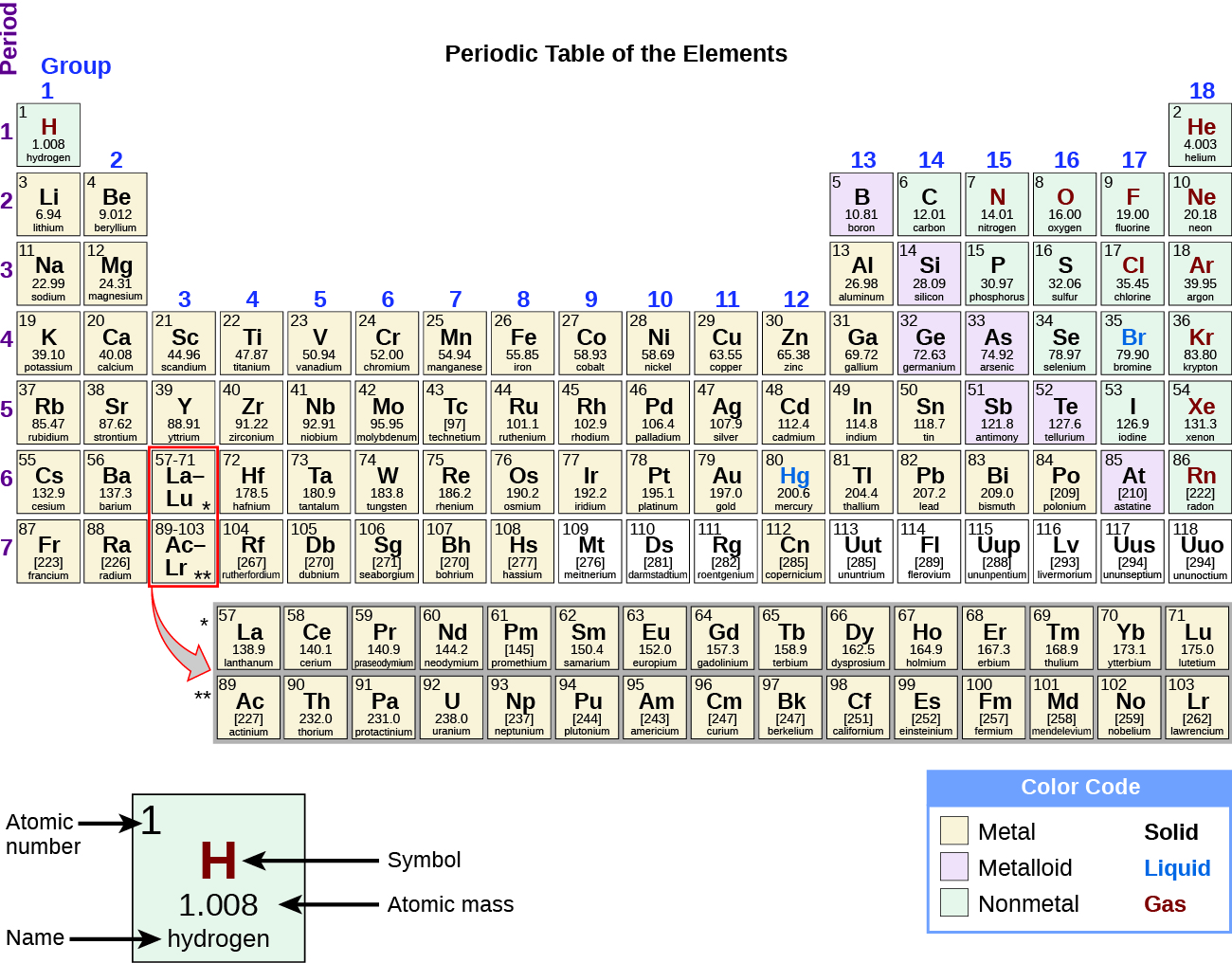
It is not safe to form hydrogen gas from sodium metal because Sodium is a highly electropositive metal and can react violently with HCL. Why is Sodium not used to prepare hydrogen? Humphry Davy was the first-ever man to isolate Sodium in 1807 by electrolysis of sodium hydroxide. It is an alkali metal in group 1 of the periodic table. Sodium with element NA is a chemical symbol and has atomic number 11. Sodium (NA) comprises 2.8% of Earth’s crust, one of the most abundant elements on Earth. It is a very soft and silvery white-colored metal. Sodium is a chemical element that belongs to the alkali metal group of the periodic table. It also contains 154 mEq/L sodium and 154 mEq/L chlorides. Sodium Chloride Injection contains nine g/L Sodium Chloride, USP (NaCl) with an osmolarity of 308 mOsmol/L. It contains 77 mEq/L and 77mEq/L chloride. How many mEq of Sodium are in normal saline? Throwing a chunk of Sodium into acid will cause an aggressive exothermic reaction until Sodium is fully consumed. Sodium is a chemically unstable metal that can react with water, air, and anything it will bind with. Why is it dangerous to add Sodium to acid? The most common of these are Potassium Compound, Sylvite and Sodium Chloride, Carnallite. It is not found in any other element of nature only as a compound. Is Chloride an element?Ĭhlorine is formed with group 17 of the periodic table known as halogens. Astatine is predicted to have a metallic crystalline structure. Antimony and Arsenic have electronic band structures made of semi-metals, although these are less stable semiconducting allotropes. Silicon, boron, tellurium, germanium are all semiconductors. When chlorine is the primary electronegative element in the compound, it is called Chloride. Chlorine helps to form compounds with oxygen and other halogens. Metal chlorides conduct electricity when dissolved or diffused in water and are decomposed by electrolysis to metal and chlorine gas. 
ADDITIONAL QUESTIONS Is Chloride a metal? The free metal cannot be found in nature naturally and needs compounds to prepare. This silvery-white, soft, and highly reactive alkali metal are in a group of 1 periodic table. Sodium NA is a chemical element and atomic number 11. If you wonder whether Sodium is a metal or nonmetal, this will clear your doubts. Like other ionic compounds containing a metal ion and nonmetal ion, Sodium Chloride is brittle and has a very high melting temperature.Ĭhlorine gas is also similar to covalent compounds, which are nonmetal with low-melting temperature. Sodium is the sixth most abundantly found element in Earth’s crust and can be found in various minerals like sodalite, feldspars, and rock salt. The free metal cannot be found in nature and needs a compound to be prepared. Sodium is of a reactive nature and is readily available in the formed hydroxide in the presence of moisture and air and cannot be used to create hydrogen. How do you calculate mEq of Sodium in normal saline?.What foods are good for low sodium levels?.Why is Sodium not used to prepare hydrogen?.How many mEq of Sodium are in normal saline?. 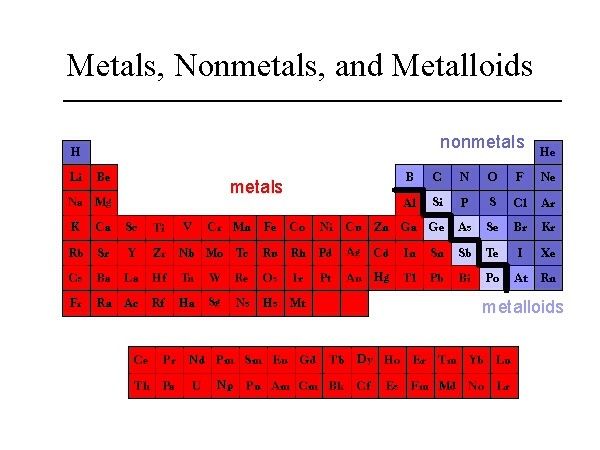
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
